Study on QTL Location of Barley / Ge Stress in Barley
Comparison of Artificial Semi-automatic and Full-automatic Phenotyping Analysis Techniques—Barley / Ge Stress QTL Location Study
Mark Crowe, Bettina Berger and Mark Tester
The Plant Accelerator, University of Adelaide Waite Campus, Hartley Grove, Urrbrae SA 5064, Australia
October 2011
design
As early as 1999, Professor Jefferies used traditional and subjective human eye observation methods to identify the phenotype of barley under boron stress conditions, and successfully obtained four QTLs related to boron stress tolerance, which controlled four different tables respectively. type. Recently, Julie Hayes and her team conducted a second independent experiment using the same QTL targeting population at the Australian Center for Plant Functional Genomics (ACPFG). In the experiment, they used germanium instead of boron to stress the barley, and used the LemnaTec automatic imaging system (similar to the Plant Accelerator) to identify and analyze the plant phenotype. Once again, they successfully verified the previously obtained QTLs and a certain phenotype. closely related. In this study, we also found that using a fully automated imaging system to identify and analyze plant phenotypes has a greater potential advantage, which includes a significant reduction in manpower input and the easy discovery of some more complex resistant QTLs And identification, etc.
background
Boron is a trace element necessary for plant growth, but higher boron content in the soil (> 1-2ppm) will cause plant toxicity, resulting in a significant decline in food crop production, which is in parts of southern Australia, Western Asia and North Africa More common. There is a genetic variation of boron tolerance in barley. Sahara 3771 has a strong tolerance to boron stress, while Clipper is more sensitive to boron stress. This study used their hybrid population as a QTL targeting population. The response of plants to boron stress is multi-faceted, which mainly includes tissue exclusion, tissue tolerance, root morphology changes, etc. The most obvious feature of barley is the gradual yellowing and necrosis of leaves from the top (Figure 1), but this is not related to tolerance Necessarily related.
The chemical structure and properties of germanium are similar to boron (and silicon), but the difference is that germanium is not a trace element necessary for plant growth, nor does it excessively inhibit plant growth under natural conditions. However, its similarity to the chemical properties of silicon is destined to play a very important role in the study of plant silicon uptake. In addition, germanium can also be transported through a boron transporter (HvNIP2; 1) in barley. This indicates that under suitable concentration conditions, it can induce symptoms similar to boron stress, and may be linked to the previously located QTL. In this study, a hybrid population of Sahara 3771 (boron-tolerant variant) and Clipper (boron-sensitive variant) was used as the QTL targeting group, and three different methods were compared: traditional human eye assessment and destructive phenotypic determination methods ; Semi-automatic destructive measurement method using LemnaTec imaging and analysis technology; It is assumed that the automatic measurement method of the plant accelerator LemnaTec system is used.

Figure 1 After 14 days of boron toxicity treatment, the leaf characteristics of the seedlings of two varieties of Sahara 3771 and Clipper with different sensitivities to boron. The germanium treatment has the same effect as the Peng treatment.
experimental design
Jefferies' experiment screened four different phenotypes: relative root length in hydroponic culture, necrotic leaf damage, tissue boron concentration, and dry matter quality. Among them, the tissue boron concentration must be destroyed even if the advanced plant accelerator imaging system is used, so this indicator is not included in the comparison, and the other three indicators can be determined or accurately estimated by image data analysis. Julie Hayes' germanium toxin test only measured leaf symptoms.
Traditional phenotyping
Relative root length
Select 150 strains from the localized population, repeat 2 times for each strain, germinate on filter paper (control: no boron solution; treatment: 100 mg / l boron solution), and then transfer to foil-coated web The medium was grown for 12 days, and the longest root was selected for determination. Relative root length (RRL) is expressed as the percentage of treated root length to control root length.
Leaf damage
The two replicates of each plant were planted in control and boron treated (100 mg / kg) soil. After four weeks, the degree of leaf necrosis was determined for each plant: 1 (no visible symptoms) -6 (> 90% necrosis).
Biomass
One week after plant leaf damage was obtained, all plants were harvested from 1 cm above ground level, dried and weighed.
For detailed steps of this phenotypic determination method, see Jefferies et al, 1999.
Semi-automatic phenotyping
Leaf damage
3-4 seedlings were selected from each strain, first cultivated by hydroponic method for 5 days, then treated with 40 μM GeO2, and continued to cultivate for 9 days. Then all the plants were taken out and placed horizontally. The LemnaTec Scanalyzer3D system was used to image and analyze the plants from above. The appropriate analysis threshold was set to record the percentage of necrotic tissue in the entire plant leaves. While imaging, the degree of leaf necrosis of each plant was observed and graded with the naked eye.
Plant accelerator phenotype determination (hypothetical)
Leaf damage, projected area (estimated biomass)
150 strains were selected from the targeted population, each strain was repeated 2 times, and then all the individual strains were cultured and boron treated according to the method in the traditional phenotyping experiment. After 5 weeks, all plants were transferred to a standard greenhouse for cultivation, and each single plant was imaged from three directions (top and two sides) using a visible light imaging unit to obtain the leaf area of ​​each plant, and then estimate its biological At the same time, the color analysis of the leaf necrosis degree of the visible light image or the chlorophyll health analysis of the fluorescence imaging is obtained.
Relative root length
The root length experiment requires an additional, boron-free control for each strain, because the root length here is relative root length and not absolute. Both control and treated plants were planted in a specific root observation pot (transparent, 400mm high, 120mm wide, 40mm deep) of the plant accelerator. The root system attached to the surface of the transparent measuring pot was imaged by visible light imaging, and the image was analyzed to obtain the plant Root length. It is worth noting that these plants can replace the plants mentioned above for the determination of biomass and leaf damage without additional plant numbers. In addition, an additional imaging can be performed when the root system has not grown to the bottom of the observation pot.
result
Traditional experiments have located 4 QTLs related to boron tolerance assessment: leaf symptom 2H chromosome QTL, relative root length 3H chromosome QTL, tissue boron concentration 6H chromosome QTL and all 4 phenotypes (the fourth type is biomass) 4TL chromosome QTL, which is consistent with the previous research results (three major gene loci linked to boron tolerance).
The image-based semi-automatic measurement experiment again found the two QTLs in the previous study, the leaf symptoms (2H) and the tissue boron concentration (6H), which is more or less strange, although different measurement methods are used . Previous studies have shown that the candidate gene for 6H QTL (boron transporter HvNIP2; 1) can also transport germanium, but there is no report that it is linked to leaf symptoms. On the other hand, although 2H QTL is closely linked to leaf symptoms, there is no evidence that it has a significant effect under boron-free conditions. More interestingly, this method did not find 4H QTL. The candidate gene for this QTL is a borate transporter and has a strong specificity. It does not have boron / germanium interaction activity like the other two QTLs. Similar to the traditional observation method, the same QTL locus can be obtained by image analysis method. There is only a small secondary peak above the main peak (see Figure 2). A gene in this region can control the flowering time, and may be different from the seedling Development speed is closely related to leaf size and shape. The secondary peak obtained by the traditional observation method is slightly larger, which indicates that the method is greatly affected by the difference in maturity, and the difference is not caused by germanium stress.
Of course, we do n’t have any data for the hypothetical fully automated plant accelerator experiment, but various circumstances indicate that with this method, we are likely to identify the above 3 QTLs, or even 6H QTL (the second test is related to leaf symptoms Closely related, but the premise of the determination is to destroy the plant leaves). In addition, through this fully automated method, we can objectively and accurately locate more QTLs, even for genes with small phenotypic changes.
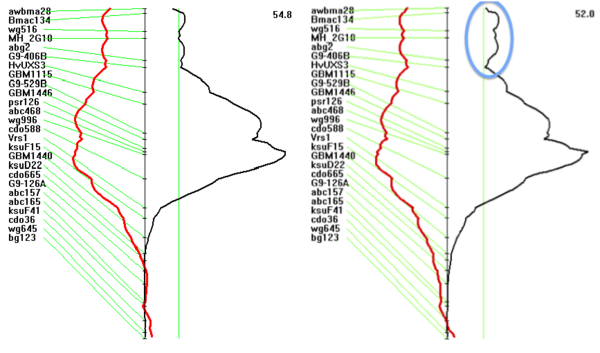
Figure 2 2H QTL linked to leaf symptoms caused by germanium stress, using LemnaTec image analysis method (left) and traditional observation method (right). The circled area highlights a secondary QTL. Compared with image analysis, the data obtained using traditional methods is more significant.
Advantages of plant accelerators
As mentioned above, even when comparing at a certain point in time, the plant accelerator can show a clear advantage. The method used by Jefferies to measure root length and leaf damage will consume a lot of time and is subjectively subjective. Even Julie Hayes said that the semi-automatic imaging method will also take two days to image the plants. Using a new type of fully automatic imaging technology will save a lot of manpower and time, and greatly increase the objectivity of the experimental data. These all indicate that the use of plant accelerator full-automatic imaging technology has advantages unmatched by other methods. The large amount of data obtained is of great significance for more in-depth discovery and identification of boron stress response QTL! In addition, the use of plant accelerator fully automatic Imaging technology can also observe and record the response of plants to boron / germanium stress in a certain period of time, which can measure root length, tissue damage and leaf area every other day, and obtain more and more reliable phenotypic data Information to find specific changes in plant phenotypes, and explore and locate more QTLs related to boron / germanium stress.
Other points to note
(1) For some simple genetic traits (such as boron stress tolerance, there are only 4 major gene loci), plant accelerator automatic imaging technology still has advantages over traditional observation methods, but some complex traits (trait expressions) may not be studied (Continuous distribution) is more obvious;
(2) Comparing the cost of the three methods, leaf symptom analysis only needs about $ 1000 (excluding the construction of the positioning population and the statistical analysis after phenotypic expression, but including manpower, consumables and greenhouse costs). However, there is no additional cost for estimating biomass using the plant accelerator fully automated imaging system, which saves almost half of the cost, and the root length measurement ($ 1000 months) is basically the same as the manual measurement.
(3)
Thanks
Thanks to Julie Hayes for the detailed description of the experimental design and digital images in this article, in-depth notes on phenotype data processing and result analysis, and at the same time, thank you Tim Sutton for his valuable comments and suggestions on this article.
references
1) Jefferies SP, Barr AR, Karakousis A, Kretschmer JM, Manning S, Chalmers KJ, Nelson JC, Islam A, Langridge P. 1999. Mapping of chromosome regions conferring boron toxicity tolerance in barley (Hordeum vulgare L.). Theoretical and Applied Genetics 98: 1293–1303.
2) Hayes J, Pallotta M, Sutton T. 2010. The relationship between boron (B) and germanium (Ge) toxicity tolerance in barley. Unpublished presentation.
3) Brenchley WE, Warington K. 1927. The role of boron in the growth of plants. Annals of Botany 41: 167–187.
4) Schnurbusch T, Hayes J, Sutton T. 2010. Boron toxicity tolerance in wheat and barley: Australian perspectives. Breeding Science 60: 297–304.
5) Cartwright B, Zarcinas BA, Mayfield AH. 1984. Toxic concentrations of boron in a red-brown earth at Gladstone, South Australia. Australian Journal of Soil Research 22: 261-272.
6) ICARDA Annual Report. 1993. Breeding winter barley for high boron soils.
7) Jenkin MJ. 1993. The genetics of boron tolerance in barley. PhD thesis, Adelaide University
8) Nikolic M, Nikolic N, Liang Y, Kirkby EA, Romheld, V. 2007. Germanium-68 as an adequate tracer for silicon transport in plants. Characterization of silicon uptake in different crop species. Plant Physiology 143: 495-503.
9) Schnurbusch T, Hayes J, Hrmova M, Baumann U, Ramesh SA, Tyerman SD, Langridge P, Sutton T. 2010. Boron Toxicity Tolerance in Barley through Reduced Expression of the Multifunctional Aquaporin HvNIP2; 1. Plant Physiology 153: 1706– 1715.
10) Langridge P, Karakousis A, Collins N, Kretschmer J, Manning S. 1995. A consensus linkage map of barley (Hordeum vulgare L.). Mol Breed 1: 389-395
11) Sutton T, Baumann U, Hayes J, Collins NC, Shi BJ, Schnurbusch T, Hay A, Mayo G, Pallotta M, Tester M, Langridge P. 2007. Boron-toxicity tolerance in barley arising from efflux transporter amplification. Science 318 (5855): 1446-1449
double chaise lounge,Chaise Lounges,leather lounge chaise
Guangzhou LoPhiDa Co.Ltd , https://www.guangdongwidinlsa.com